Deconstructing the Opioid Crisis
OxyContin serves as a powerful example of how corporate interest can drastically shape medical practice and health worldwide.
Read Time: 4 minutes
Published:
In the United States, patients are frequently discharged with prescription opioids for pain management following surgery, including routine procedures like wisdom-teeth removal. Medications like Percocet and OxyContin promise relief during the healing process, yet have also fueled one of the most deadly public health crises in history. Purdue Pharma introduced OxyContin in 1996 and reshaped the landscape of pain management, catalyzing what we know as the global opioid crisis today.
The U.S. Food and Drug Administration (FDA) approved OxyContin as a safe treatment for moderate-to-severe pain, in addition to severe pain related to injury or cancer. The broad approval allowed Purdue to market the drug for conditions that do not typically warrant powerful opioids. Executives deemed addiction “very rare” due to the extended-release formulation, and began promoting OxyContin as a long-acting pain reliever.
The Sackler family, who have owned Purdue since the 1950s, carefully orchestrated the commercial success of OxyContin. They invested heavily in marketing directed at doctors, pain advocacy groups, and medical journals. Sales representatives were also incentivized by annual bonuses to encourage doctors to prescribe OxyContin. Their strategy proved highly successful, and by 2001, Purdue was earning more than $1 billion annually. As profits and prescriptions surged across the country, so did rates of misuse and overdose.
The crisis hit the western United States the hardest, pervading rural and ethnic communities in West Virginia and New Hampshire. To understand the extent of Purdue’s influence, Andrea Bowra and colleagues conducted a case study examining 36 public documents related to the global health response to the opioid crisis. Using actor-network theory, the researchers mapped relationships among organizations that were involved in opioid policy and response efforts between 2007 and 2022.
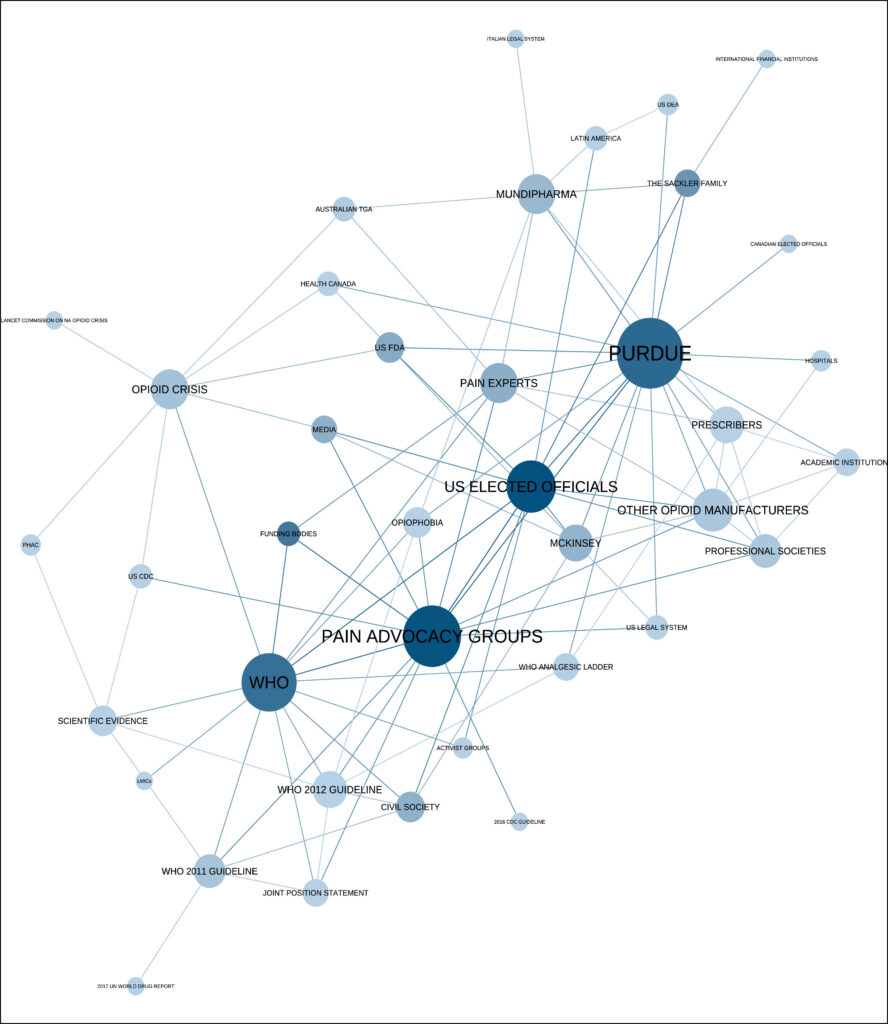
As shown in the figure, the analysis identified 39 actors (circles) and 99 relationships (lines) that shaped global responses. Purdue Pharma emerged as a central actor, with connections to advocacy groups, policymakers, and international organizations.
One major strategy that Purdue used to garner support was “astroturfing,” or creating fake grassroots groups that appeared to operate independently. In one instance, Purdue privately funded the American Pain Society to advocate against strict prescribing guidelines and for broader opioid use. Lobbyists from the group influenced the FDA to support the expansion of OxyContin, including the approval of its use in children without strong evidence of safety.
Purdue Pharma also shaped how medical professionals used official pain management tools. The World Health Organization (WHO) originally designed the 3-step analgesic ladder for cancer pain, advising doctors to prescribe non-narcotics before opioids. As opioids became systematically normalized over time, Purdue executives influenced the WHO to stray from the ladder and encourage more frequent opioid use.
OxyContin serves as a powerful example of how corporate interest can drastically shape medical practice and health worldwide.
At the same time, Purdue coined and popularized the term “opiophobia” in its marketing campaigns. The term reframed concerns about addiction into an “unreasonable fear” from reluctant doctors, effectively creating stigma among prescribers. The researchers also identified key policymakers in the network, specifically 23 U.S. elected officials who helped pass policies to limit opioid regulation. Drug companies contributed over $1.5 million to the campaigns of lawmakers who backed legislation that restricted investigations into suspicious narcotic activity.
Similar deep-rooted connections allowed Purdue and the Sackler Family to sustain OxyContin sales during the peak of the crisis. While evidence indicates the Sacklers knew about OxyContin’s potential for abuse back in 1997, they continued marketing and selling the drug for two more decades. After amassing thousands of lawsuits from victims for deceptive marketing and improper sales tactics, Purdue Pharma filed for bankruptcy and pleaded guilty to felony charges in 2020. Separately, the Sacklers agreed to a $7.4 billion settlement as restitution to the victims of the opioid crisis, but they have yet to face criminal charges for their involvement.
More than two decades after its launch, OxyContin serves as a powerful example of how corporate interest can drastically shape medical practice and health worldwide. Money from the settlement has been used to pay for addiction treatment, counseling, and housing for thousands of victims still in recovery. The authors emphasize transparency in the pharmaceutical industry to prevent corrupt ties among drug companies, policymakers, and doctors from influencing care. Continuing to heal from the opioid crisis requires increasing access to evidence-based pain management alternatives, stricter pharmaceutical oversight, and rebuilding trust across the globe.



